Cancer chemotherapy is usually associated with a wide range of devastating side effects that seriously impair cancer survivors' quality of life (Ahles and Saykin, 2007). Chemotherapy-induced cognitive impairment (CICI), also known as chemobrain or chemofog, is the most detrimental adverse effect manifested by weakened memory, reduced learning and processing speed, and decreased attention either throughout or following chemotherapy (Koppelmans et al., 2012; Lange et al., 2019). Chemofog has been attributed to the tissue damage triggered by mostly the oxidative insult, neuroinflammation, and mitochondrial disruption; the exact underlying mechanism has yet to be fully understood. 75% of cancer patients suffer from this neurological disorder during chemotherapy (Flanigan et al., 2018; Shi et al., 2019). The majority of CICI cases have been reported among breast cancer-afflicted women undergoing chemotherapy (Jim et al., 2012).
Doxorubicin (DOX) is a well-known drug that is broadly utilized as a remedy for cancers of the breast, thyroid, prostate, ovary, and Hodgkin’s lymphoma. It is a member of the anthracycline group, which displays anticancer activity via 3 ways: interfering with DNA function, impacting the growth of cancer, blocking topoisomerase, releasing excessive free radicals (El-Agamy et al., 2019). These mechanisms destroy the essential biomolecules, thereby leading to cancer cell death. Unfortunately, the cytotoxic actions of DOX damage the healthy cells as well, causing toxicities of different organs such as cardiotoxicity, hepatotoxicity, nephrotoxicity, and neurotoxicity (Carvalho et al., 2009). Tangpong et al. (2006) and Ongnok et al. (2020) revealed that DOX can cause significant damages in the brain through a secondary mechanism by increasing peripheral cytokine tumor necrosis factor alpha (TNF-α) levels which penetrates the blood brain barrier (BBB) and impairs antioxidant defense, resulting in synaptic dysplasticity, death of neurons, and ultimately cognitive dysfunction.
Chemofog is associated with a reduction in neurotrophins concentrations which are necessary for neuron growth and survival (Saral et al., 2021). Brain-derived neurotrophic factor (BDNF) is one of the neurotrophins found in the brain with abundant expression in the hippocampus (Murer et al., 2001). It possesses neuroprotective properties as it participates in the regulation of glial cells and neurons proliferation and in the modulation of synaptic plasticity of both long- and short-term memories (Kowianski et al., 2018). Park et al. (2018) reported that the decrease in BDNF and its receptor, tropomyosin receptor kinase B (TrkB) accounted for the attrition of the recently formed neuronal cells in the hippocampus, resulting in the disturbance of working memory and spatial learning after administration of DOX. Currently, there is no effective and safe therapy for chemofog in cancer survivors. Numerous researches have highlighted the possible role of phytochemicals in ameliorating a variety of neurodegenerative conditions (Beking and Vieira, 2019; Maher, 2019). For instance, curcumin exhibited neuroprotective effect in parkinson’s disease model via upregulating Trk/phosphoinositide 3-kinases (PI3K) signaling and elevating BDNF levels (Yang et al., 2014). In addition, berberine has been proven to ameliorate cognitive disturbances caused by DOX via enhancing BDNF through activating SIRT-1 (Shaker et al., 2021).
Among several biologically active phytochemicals, Bisabolol (BISA), also called levomenol, has garnered considerable interest owing to its diverse pharmacological features, including anti-infective, anti-oxidant, anti-inflammatory, anti-nociceptive, gastroprotective, cardioprotective, and anti-cancer activities (Braga et al., 2009; Rocha et al., 2011; Forrer et al., 2013; Barreto et al., 2016; Meeran et al., 2020). It is a monocyclic sesquiterpene alcohol that was initially isolated from Matricaria chamomilla plant (Mckay and Blumberg, 2006). BISA is found in nature in two distinct forms, ά and β. The ά-isomer form is responsible for the majority of its biological characteristics. Owing to its high lipophilicity, BISA can easily undergo oxidation and generate two bisabolol oxides (A and B) (Arunachalam et al., 2022). Earlier studies have elucidated the palliative effect of BISA against cardiac and renal toxicity caused by DOX (Arunachalam et al., 2022; Meeran et al., 2023), cadmium-induced liver injury (Elazab and Hsu, 2022), neurodegeneration in rotenone (ROT)-provoked rat model of Parkinson’s disease (Javed et al., 2020), and neuronal injury and memory disorders caused by cerebral ischemia in mice (Fernandes et al., 2019). The neuroprotective effect of BISA against cognitive disturbances caused by DOX has not been explored. However, other sesquiterpenoids, e.g., β-caryophyllene have exhibited abrogative action against cognitive disorder to both long- and short-term memory caused by aspartame via enhancing the BDNF/TrKB signaling pathway (Rosa et al., 2023).
The present study was designed to assess the potential ameliorative action of BISA against DOX-induced cognitive dysfunction and the mechanisms implicated in this action. We hypothesize that BISA’s palliative effect on DOX-induced chemofog is through its antioxidant, anti-inflammatory, and anti-apoptotic properties, as well as its effect on the BDN/TrKB pathway.
2 Materials and methods2.1 ChemicalsDoxorubicin injectable solution (2 mg doxorubicin hydrochloride/mL) was supplied from EBEWE Pharma Ges.m.b.H. Nfg. KG, Unterach am Attersee, Austria. α-Bisabolol (BISA, Cas No. 23089-26-1) was purchased from Sigma Aldrich Co. (St. Louis, MO, United States). BISA was prepared with sunflower oil (50 and 100 mg/mL for administration to rats at doses 50 and 100 mg/kg, respectively).
2.2 AnimalsThis research was carried out on 60 male Wistar rats (180–200 gm) obtained from the Laboratory Animal House (Zagazig University, Egypt). Rats were kept in plastic cages in well-controlled housing conditions with a temperature 24°C ± 1°C, humidity 60%–70%, and alternating shifts of 12 h dark and 12 h light. They were supplied with food pellets and drinking water ad libitum and were left to adapt to the experimental environment for 7 days before the commencement of the investigation. The procedures of this study were revised and authorized by the Mansoura University Animal Care and Use Committee.
2.3 Sample size calculationSample size was calculated depending on a former research by Egwuatu et al. (2023) employing G*power 3.1.9.4 software, according to the difference between two independent study groups (control Vs. DOX groups) utilizing t-test with a probability type I error (α) = 0.05 and a power 0.9. The sample size was estimated to be 30 rats (6 rats/group). A larger number of rats (12 rats/group) was enrolled in each experimental group at the beginning of the study as we expected that there will be mortalities in the groups receiving DOX and that some rats may be excluded in the training session of the passive avoidance test (those who failed to step into the dark chamber in the training session within 90 s).
2.4 Experimental designThe examined rats were randomly assigned to 5 groups (12 rats/group). Group I (control) was injected with normal saline (0.2 mL/rat, vehicle for DOX) intraperitoneally (i.p.) once/week at 0, 7, 14, 21 days of the trial and was administered sunflower oil (BISA vehicle) at 1 mL/kg/day orally for 4 weeks. Group II (BISA group) received normal saline i. p. once a week and was given BISA in sunflower oil by gavage at 100 mg/kg/day for 4 weeks. Group III (DOX group) was injected i. p. with DOX at 2 mg/kg/week (El-Agamy et al., 2018; Shaker et al., 2021) and was given sunflower oil orally every day for 4 weeks. Groups IV and V were administered DOX at 2 mg/kg i. p. weekly and BISA orally at 50 and 100 mg/kg/day, respectively, for 4 weeks. The doses for BISA were selected based on published reports (Javed et al., 2020; Fernandes et al., 2019; Cavalcante et al., 2020; Nazarinia et al., 2023).
2.5 Behavioral tests2.5.1 Passive avoidance testA passive avoidance test was conducted to examine the short-term memory as illustrated by Abdel-Aziz et al. (2016) using the apparatus (Passive Avoidance Set-up, Ugo Basile, Italy). This device had two compartments, one was illuminated by a 10-W lamp, whereas the second chamber was dark with a grid floor that was set to shock the rats with an electric shock at a certain intensity when they passed on it. An automated sliding door connects these two chambers. The experiment was performed in two sessions for each rat; training and test one. In the training period (performed on the 28th day of the study), each rat was smoothly put in the lit compartment, and when the rat entered the black chamber, placing its feet on the floor, the door closed, and an electric shock (1 mA) was directed for 2 s for each animal. Rats that did not enter the dark chamber within 90 s were excluded from the study. One day after the training session, the test session was carried out (on the 29th day of the experiment). Rats were set in the lighted chamber, and the time taken to enter the dark chamber (known as the step-through latency) was monitored, and this time is regarded as a step-through reaction to assess the rat’s memory acquisition following the exposure to a painful stimulus. A cutoff period of 300 s was considered in the test, and rats were not subjected to electric shock in the dark chamber during the test session.
2.5.2 Locomotion assessmentTo exclude the influence of motor disorders on the step-through latency of passive avoidance test, the locomotor activity of rats was evaluated employing an animal activity detector (Opto-Varimax-Mini Model B; Columbus Instruments, OH, United States). Rat movements inside this device led to interruptions to infrared beams (scan rate = 160 Hz, wavelength = 875 nm, diameter = 0.32 cm, and spacing = 2.65 cm) that were counted. According to El-Agamy et al. (2018), rat’s locomotor activity was determined as counts/5 min.
2.5.3 Morris water maze test (MWM) testFor assessing the learning ability and spatial memory, MWM test was performed as described by Jang et al. (2013). We used MWM apparatus with a circular pool (180 cm in diameter, 60 cm in height, containing water at 25°C–27°C). This pool was divided into 4 identical quadrants; one of them contains a concealed escape platform at 2 cm water depth. All animals were trained 3 times/day with platform position for 4 days. During each training trial, rats were allowed to swim from various release positions and trained to get into the concealed platform, where they were directed to remain there for 30 s prior to placing them in their cages. The time spent by each rat to find the platform (latency time) was registered. Rats that were unable to arrive at the platform within 90 s, were manually directed to it. After the last training session (on the fifth day of the test), animals were subjected to the probe trial test where the platform was eliminated and the rats were permitted to search for the eliminated platform for up to 90 s and the period spent by the animals in the platform quadrant was documented.
2.6 Samples collectionOn the 32nd day of the experiment (at the end of behavioral tests after finishing MWM test), 6 rats from each group were cervically decapitated. Then, the entire brain from each rat was promptly excised, and the hippocampus was removed and divided into 3 portions. The first portion underwent homogenization process followed by centrifugation at 3,000g for 10 min, and the collected supernatant was utilized for investigating the redox status. The second portion was immersed in 10% formalin for the preparation of paraffin blocks required for microscopic examination of hippocampal tissues. The last portion was frozen at −80°C for gene expression assessment and enzyme-linked immunosorbent assay (ELISA).
2.7 Redox status in hippocampal tissuesBy using spectrophotometer, the level of reduced glutathione (GSH), superoxide dismutase (SOD) and catalase activities (CAT) in the homogenate supernatant of hippocampal tissues were measured as described previously by Beutler (1963), Nishikimi et al. (1972), and Fossati et al. (1980), respectively. In addition, the technique of Ohakawa et al. (1979) was employed to evaluate malondialdehyde (MDA) content.
2.8 ELISAELISA was utilized to estimate the hippocampal levels of interleukin-1β (IL-1β), IL-6 (Bioassay Technology Laboratory Co. Shanghai, China), total Nuclear factor kappa B (NF-kB, the total protein level from both nuclear and cytoplasmic fractions), acetylcholinesterase activity (AChE), concentrations of Gamma-aminobutyric acid (GABA) (Fine Test Co., Boulder, CO, United States), acetylcholine (Ach, Elabscience Co. Houston, TX, United States), serotonin (5-HT, AFG Scientific, Wood Dale, IL, United States), dopamine (DA, Cusabio Co., Houston, TX, United States), glutamate (Novus Biologicals Co., Centennial, Co., United States), BDNF (Biosensis Pty Ltd., Thebarton, SA, Australia), and TrkB (Antibodies co. Cambridge, United Kingdom). The guidelines issued by the manufacturers were followed when using these ELISA kits.
2.9 Detection of the transcription levels of Nrf2, HO-1, Bax, and Bcl-2, P38 MAPK, and SIRT-1 genes in hippocampus by qRT-PCRAs per the directions recommended by the manufacturer, the QIAamp RNeasy Mini kit (Qiagen Inc., MD, United States) was implemented to isolate the total RNA from the hippocampal specimens of all experimental rats. The extracted RNA’s purity and concentration were assessed by a NanoDrop (UV-Vis spectrophotometer Q5000, United States). Afterwards, reverse transcription of RNA to cDNA was accomplished utilizing SensiFast™ cDNA synthesis kit (cat. No. Bio-65053, Bioline Ltd., United Kingdom). The mRNA levels of Nuclear factor erythroid 2-related factor (Nrf2), Heme oxygenase-1 (HO-1), Bax, and B-cell lymphoma 2 (Bcl-2), P38 mitogen-activated protein kinase (P38 MAPK), and Sirtuin-1 (SIRT-1) genes were evaluated with the aid of a Stratagene MX3005P real-time PCR machine (Agilent, CA, United States) employing SYBR Green PCR Master Mix (2x SensiFast™ SYBR, Bioline Ltd., United Kingdom). To normalize the expression of the examined genes, β-Actin, a reference housekeeping gene, was utilized. Table 1 displays the sequences of the used primers for the target genes. The qRT-PCR reaction was achieved under the following conditions: starting with initial denaturation at 94°C for 15 min (40 cycles), with subsequent heat activation for 15 s at 94°C, primers annealing at 68°C (30 s) for Bcl-2, 58°C (30 s) for P38 MAPK, and at 60°C (30 s) for the other remaining tested genes, and finally extension temperature was set at 72°C for 30 s. The 2−ΔΔCT (Ct: cycle threshold) calculation was applied for relative estimation of the transcription levels of the analyzed genes (Yuan et al., 2006).
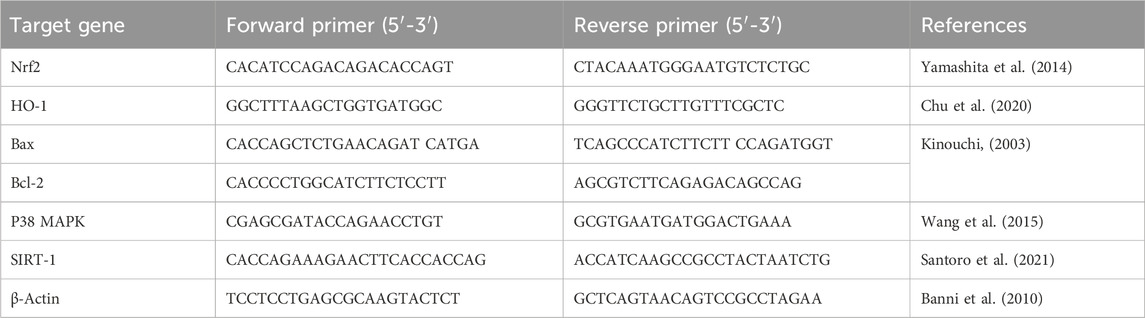
Table 1. The sequences of primers utilized for quantitative real-time PCR investigation.
2.10 Microscopic examination of hippocampal samples2.10.1 Hematoxylin and eosin (H&E) stainingAfter preservation in 10% formalin, the hippocampal tissue samples of all groups were dehydrated with ascending grades of alcohol, cleared using xylene, and submerged in paraffin wax. Thereafter, a microtome was employed to slice 4 µm thick tissue sections, which were assembled on glass slides. The slides were scrutinized under a light microscope after staining with H&E as delineated by Suvarna et al. (2018). The degenerated neurons were quantified in CA3 and dentate gyrus from the slide of each rat for 6 rats/group. The grading and quantitative scoring of neuronal degeneration was conducted following the method of Khalil et al. (2022) [scale score: 0 = normal; 1 = mild (<25%, scattered neurons); 2 = moderate (25%–50%, scattered neurons); 3 = sever (>75%, scattered neurons); 4 = sever, multifocal]. The degenerated neurons were identified based on cell morphology, cytoplasmic appearance, nuclear condensation, and neurofibrillary integrity.
2.10.2 Toluidine blue stainingAdditional slides for the hippocampus were impregnated with 0.1% toluidine blue for 2 min. After dehydration of the sections, they were mounted on Canada balsam, and the slides were visualized by a microscope (Bancroft and Gamble, 2008). The neurons in the hippocampus were regarded as viable if they have round nuclei and observable nucleoli. In contrast, highly stained and shrunken cells indicate degenerated neurons which were counted in CA3 and dentate gyrus from every slide of each rat for 6 rats per experimental group. The number of damaged neurons was reported as a ratio out of 100 neurons in each area.
2.10.3 ImmunohistochemistryTNF-α and glial fibrillary acid protein (GFAP) immunohistochemical staining in the hippocampal sections was carried out according to the description of Khafaga et al. (2021). Briefly, the deparaffinized sections were rehydrated utilizing ethanol at descending concentrations. After unmasking the antigen by boiling for 10–20 min at 105°C, the slides were rinsed with phosphate buffer saline (PBS). Then, the slides were dipped in 3% H2O 2 (in pure methanol) for 5 min at 25°C to suppress the endogenous peroxidase. Thereafter, 10% normal goat serum was added and maintained for 1 h to abolish the non-specific reaction. The prepared hippocampal slides were subjected to 12 h incubation with the primary antibody against TNF-α (rabbit polyclonal IgG, 1: 200 dilution, cat. No. AP20373PU-N, Acris, Germany) and GFAP (rabbit polyclonal IgG at 1:200 dilution, cat. No. ab7260, Abcam, Cambridge, United Kingdom) at 4°C. Afterwards, the sections underwent incubation for 60 min with biotin-conjugated goat anti-rabbit IgG (Histofine kit, Nichirei Corporation, Japan) and for 30 min with streptavidin-peroxidase conjugate (Histofine kit, Nichirei Corporation, Japan). Diaminobenzidine (DAB) chromogen was added to inspect the streptavidin-biotin reaction. After counterstaining with hematoxylin, the slides were visualized by a pathologist using a light microscope.
The brown zones were measured as optical density (OD) in cornu ammonis 3 (CA3) and dentate gyrus of each rat for six rats per group employing an ImageJ software (National Institutes of Health, Bethesda, 150 MD, United States).
2.11 Statistical analysisThe statistical assessment of all data was executed by applying Graphpad Prism 5 (Graphpad Software, CA, United States). The Shapiro-Wilk approach was utilized to scrutinize the normality of the data. The numerical data were expressed as mean ± SEM. Two-way ANOVA followed by the Bonferroni post hoc test was implemented to analyze the data for the training session of the MWM test (escape latency data). Meanwhile, all other remaining results were statistically computed employing one-way ANOVA followed by Tukey’s post hoc test. The statistical significance threshold was set at p < 0.05.
3 Results3.1 Effect of BISA on DOX-induced cognitive disordersNo remarkable difference was recorded in the step-through latency among the tested groups throughout the training period of the passive avoidance experiment (Figure 1A). Throughout the test period, DOX administration significantly reduced (p < 0.05) the step-through latency by 75% relative to the control rats, indicating memory deterioration. BISA at 50 and 100 mg/kg/day along with DOX dose-dependently increased (p < 0.05) the step-through latency in comparison with those exposed to DOX alone (Figure 1B).
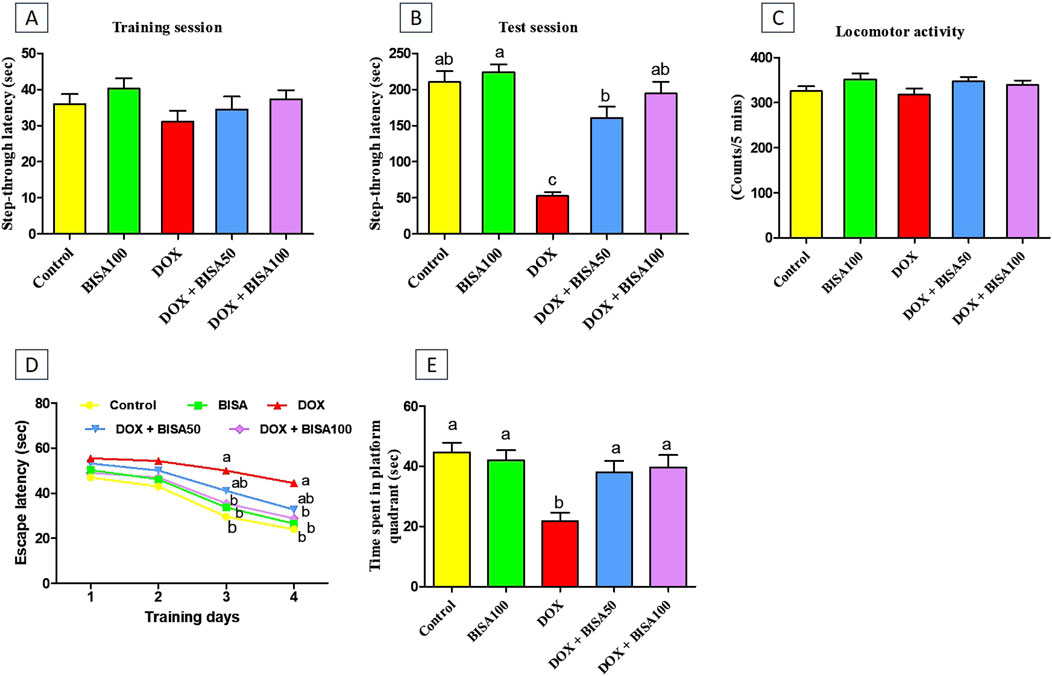
Figure 1. Impact of α-bisabolol on behavioral alterations caused by doxorubicin in rats. (A) Step-through latency (training session), (B) Step-through latency (test session), (C) Locomotor activity, (D) Morris water maze test (MWM, escape latency): 4 training days, (E) MWM (time spent in platform quadrant). All data were exhibited as mean ± SEM (n = 6). BISA50, BISA100: α-bisabolol was given by gavage at 50 and 100 mg/kg, DOX: doxorubicin. The findings for (D), which represent the mean escape latency were statistically assessed by applying two-way ANOVA followed by Bonferroni post hoc test. Within each training day, different letters highlighted statistical variation (p < 0.05). Meanwhile, statistical investigation of other behavioral data was carried out employing the one-way ANOVA, then Tukey’s post hoc test. Bars designated with various lowercase letters are significantly different (p < 0.05).
The locomotor activity was evaluated for all rats to preclude the influence of motor disorders on the outcomes of all executed neurobehavioral tests including the passive avoidance test. As displayed in Figure 1C, there were no statistical differences in the locomotor function among all groups.
The MWM test was performed to assess the learning and spatial memory of all rats. In the first 2 days of the training, all groups showed no significant difference in the mean escape latency time. DOX-treated rats exhibited lower capability to reach the hidden platform with a marked increase (p < 0.05) in escape latency time on the third and fourth days of the training session compared to the control group. In contrast, supplying DOX-treated rats with BISA, particularly at 100 mg/kg/day, resulted in significant reduction (p < 0.05) in the escape latency time compared to the DOX group (Figure 1D). On the fifth day (probe trial test), rats receiving DOX only exhibited a significant decrease (by 51%) (p < 0.05) in the time spent on the target platform quadrant in comparison with the controls. Conversely, the concurrent treatment with BISA (DOX + BISA50 and DOX + BISA100 groups) significantly increased (p < 0.05) the time spent on the target platform in a dose-dependent manner as compared to the DOX group (Figure 1E).
3.2 Effect of BISA on DOX-provoked hippocampal oxidative insult and on the mRNA levels of Nrf2 and HO-1 genesDOX administration triggered a vigorous state of oxidative insult in the hippocampus manifested by a significant elevation (p < 0.05) in the content of lipid peroxidation marker (MDA) by 209% and a drastic decline (p < 0.05) of CAT, SOD and GSH levels (by 68%, 55%, and 60%, respectively) compared to the control group. Co-administration with BISA (at 50 and 100 mg/kg/day) reduced MDA levels and increased the CAT, SOD, and GSH levels in a dose-dependent manner. The DOX + BISA100 group showed better ameliorative effect on the redox status, where these oxidative insult biomarkers were restored to the control levels (Table 2).
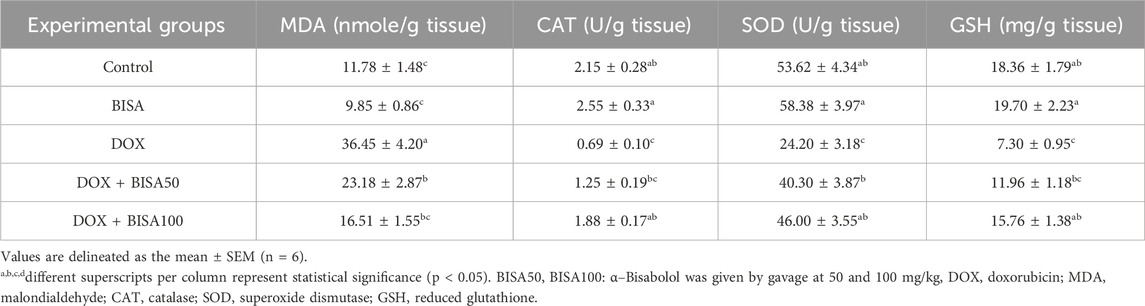
Table 2. Effect of α-Bisabolol on DOX-induced oxidative insult indices in hippocampal tissues of experimental rats.
Furthermore, BISA caused a significant upregulation (p < 0.05) of Nrf2 gene and its antioxidant response element HO-1 in DOX + BISA50 and DOX + BISA100 groups compared to DOX group (Figures 2A,B).
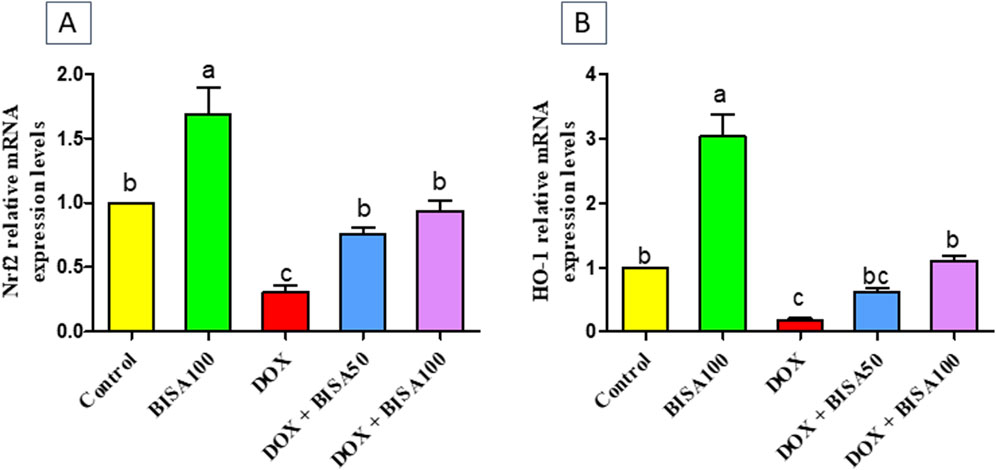
Figure 2. Impact of α-bisabolol on the transcription levels of Nrf2 (A) and HO-1 (B) in DOX-induced cognitive impairment in rats. Values are displayed as mean ± SEM (n = 6). The letters on every bar point out statistical significance between them (p < 0.05). BISA50, BISA100: α-bisabolol was administered orally at 50 and 100 mg/kg, DOX: doxorubicin was injected i. p. Once/week for 4 weeks, Nrf2: nuclear factor-erythroid 2-related factor-2, HO-1: heme oxygenase-1.
3.3 Effect of BISA on DOX-induced histopathological alterations in the hippocampusThe control and BISA groups showed normal neuronal configurations in CA3 and dentate gyrus regions appeared as normal round intact neurons with normal lightly stained nucli, and no obvious degeneration was noticed. While the DOX-treated group displayed severe neuronal degeneration in these 2 regions observed as neuronal shrinkage, more eosinophilic cytoplasm and pyknotic nuclei besides vacuolations and neurofibrillar loss noticed only in CA3. Concurrent administration of BISA at 50 mg/kg/day with DOX reduced the neuronal degeneration in both regions. Moreover, the slides obtained from DOX + BISA100 group exhibited mild degeneration (<25%, scattered degenerated neurons) in CA3 and dentate gyrus areas. The quantitative data for these histological changes revealed statistical significance (P < 0.05) in the scores of degenerated neurons between DOX, DOX + BISA50, and DOX + BISA100 groups (Figures 3A,B).
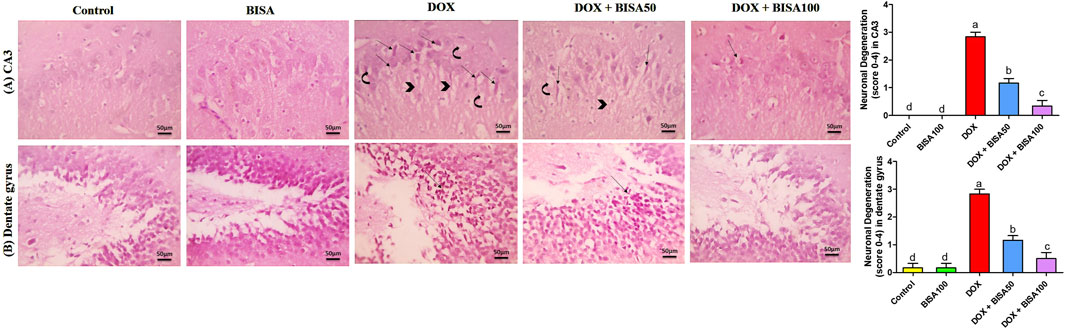
Figure 3. Microscopic photos of hippocampal slides from rats stained with H&E. (A) Hippocampal cornu ammonis 3 (CA3) sections, (B) hippocampal dentate gyrus sections. Slides from control and BISA100 (α-bisabolol oral administration at 100 mg/kg/day) groups exhibited no neuronal degeneration in both regions (CA3 and dentate gyrus). On the other hand, sections from the group treated with doxorubicin only (DOX group) revealed many degenerated neurons (thin arrows) with vacuolations (curved arrow) and neurofibrillar loss (arrowheads) in CA3 region and severe neuronal degeneration (thin black arrow) in dentate gyrus region. Tissues from DOX + BISA50 displayed fewer degenerated neurons (thin arrows) with vacuolations (curved arrow) and neurofibrillar loss (arrowhead) in CA3 region and showed decreased neuronal degeneration (thin black arrow)in dentate gyrus area. Hippocampal sections CA3 from DOX + BISA100 group exhibited few degenerated neurons (thin arrows), while denatte gyrus region from the same group revealed normal neurons. X400 bar 50 μm.
These results were emphasized using toluidine blue stain. As shown in Figures 4A,B, CA3 and dentate gyrus regions from control and BISA100 groups revealed rounded neuronal build, normal cytoplasm, well-defined nuclei. In contrast, the two regions in the DOX-group exhibited prominent neuronal degeneration as indicated by loss normal rounded framework of normal neurons with darkened cytoplasm, hyperchromatic and pyknotic nuclei. DOX + BISA 50 group showed decrease in the number of degenerated neurons in CA3 and dentate gyrus regions and much more reduction was observed in DOX + BISA100. The percentage (%) of degenerated neurons in CA3 and dentate gyrus regions was shown in Figures 4A,B.
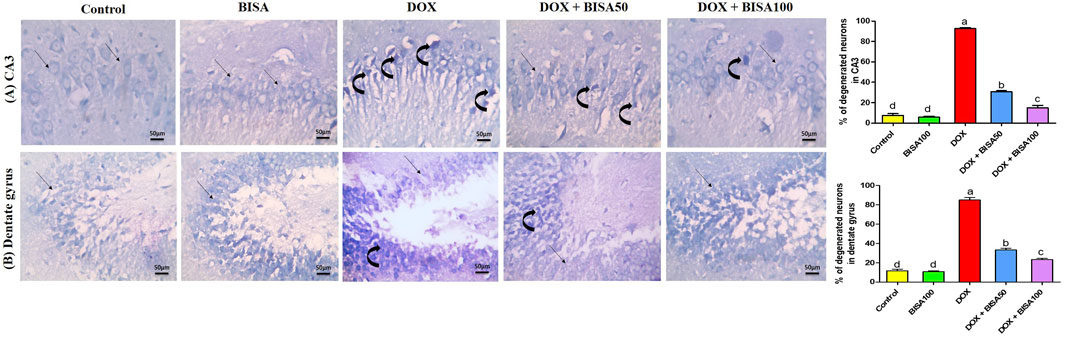
Figure 4. Photomicrographs of hippocampal sections stained with toluidine blue (X 400 bar 50 μm). (A) Hippocampal cornu ammonis 3 (CA3) sections, (B) hippocampal dentate gyrus sections. Control and BISA100 (α-bisabolol was gavaged orally at 100 mg/kg/day) groups showed normal blue-stained neurons (thin black arrow) in both regions of hippocampus (CA3 and dentate gyrus). On the contrary, sections from DOX-exposed rats showed many dark stained neurons (curved arrows) in the two regions. Slides from DOX + BISA50 presented fewer dark stained neurons (curved arrow) in the two hippocampal regions. DOX + BISA100 group showed many normal blue-stained neurons (thin black arrow) with few dark stained neurons (curved arrows) in CA3. The number of degenerated neurons was expressed as a percentage from 100 neurons in each area. Each bar labeled with various letters is significantly different (p < 0.05). Data are exhibited as mean ± SEM (n = 6).
3.4 Impact of BISA on DOX-induced neuroinflammation and astrogliosis in the hippocampusThe BISA100 group had no remarkable alterations in the hippocampal concentrations of the inflammatory markers including NF-kB (Figure 5A), IL-1β (Figure 5B), and IL-6 (Figure 5C). DOX administration provoked robust inflammatory reactions mirrored by the marked upsurge (p < 0.05) in the levels of these inflammatory indices in comparison with the control group. However, groups treated with DOX and BISA concurrently (DOX + BISA50 and DOX + BISA100 groups) showed a considerable reduction (p < 0.05) in the concentrations of these markers as compared to the rats receiving DOX alone. Moreover, no significant difference in the levels of NF-kB, IL-1β, and IL-6 was observed between the DOX + BISA100 group and the control group.
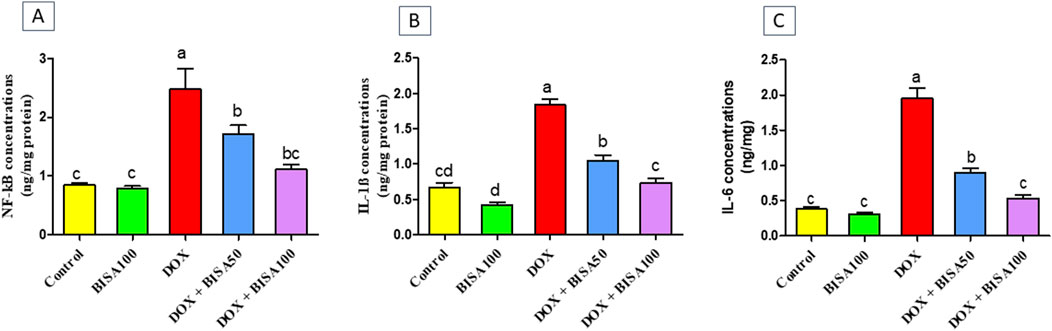
Figure 5. Effect of α-bisabolol on the concentrations of inflammatory markers detected by ELISA in hippocampus tissues of DOX-subjected rats. (A) NF-kB, (B) IL-1β, (C) IL-6. The results are shown as mean ± SEM (n = 6). Bars with different letters are substantially varied from one another (p < 0.05). BISA50, BISA100: α-bisabolol was offered at 50 and 100 mg/kg/day per os, DOX: doxorubicin was given i. p. One injection every week for 4 weeks, NF-kB: Nuclear factor kappa B, IL-1β: interleukin-1β, IL-6: interleukin-6.
The immunohistochemical investigation of the pro-inflammatory cytokine TNF-α revealed negative expression in the CA3 and dentate gyrus regions of the control and BISA100 groups. In contrast, a brown neuronal TNF-α expression was seen in these two regions of the rats treated with DOX only. Notably, concomitant administration of BISA at 50 mg/kg/day led to a reduction in the positive TNF-α staining in CA3 and dentate gyrus. Additionally, mild positive expression was detected in the tissues collected from rats treated with the higher dose of BISA (100 mg/kg/day) along with DOX. Quantitation of TNF-α staining was evaluated as optical density of the immunostained areas (Figures 6A,B).
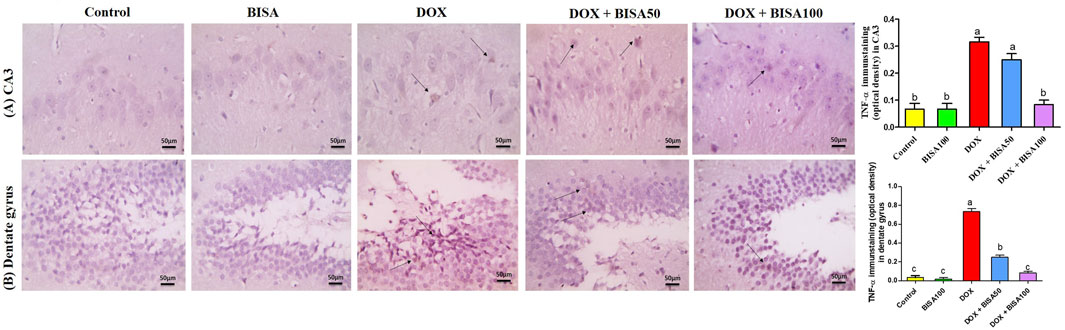
Figure 6. Histological images of immunostained hippocampal slides against tumor necrosis factor α (TNF-α) (X400 scale bar 50 μm). (A) Sections for hippocampal cornu ammonis 3 (CA3) region, (B) sections for hippocampal dentate gyrus region. Images from the control and BISA100-exposed rats revealed negative TNF-α expressions in CA3 and dentate gyrus. In contrast, DOX group showed positive brown staining for TNF-α in both examined areas of hippocampus (thin black arrow). The slides obtained from rats in DOX + BISA50 group displayed decrease in the positive brown staining for TNF-α in the two hippocampal areas (thin black arrow). DOX + BISA100 group presented mild positive brown TNF-α expression in the neurons in both screened regions (thin black arrow). The intensity of brown areas (TNF-α expression) in the experimental groups was exhibited as optical density (OD). Values were depicted as mean ± SEM (n = 6). There is a significance variation (p < 0.05) among bars that convey letters (p < 0.05).
The immunohistochemical analysis of GFAP in the hippocampus displayed a few astrocytes in CA3 and dentate gyrus of the control and BISA100 groups. Multiple highly branched astrocytes were detected in the hippocampal sections of the DOX-treated rats. However, the slides from DOX + BISA50 group showed fewer less branched astrocytes in these two regions. Also, the DOX + BISA100 group showed very few astrocytes in these two regions. Quantitative assessment of immunohistochemcial staining for GFAP was represented as optical density (Figures 7A,B).
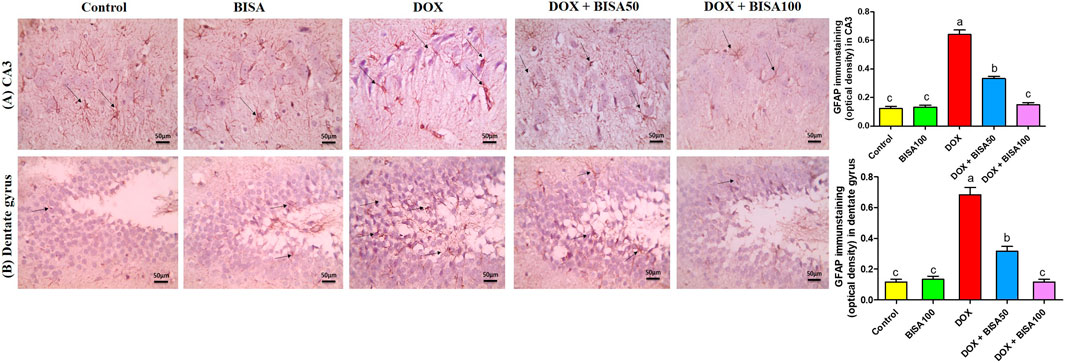
Figure 7. Immunohistochemical staining pictures for glial fibrillary acid protein (GFAP) in the hippocampus of experimental rats (X 400 scale bar 50 μm). (A) Images for cornu ammonis 3 (CA3) region, (B) images for dentate gyrus region. The sections from the control and BISA100 group exhibited few astrocytes in CA3 and dentate gyrus zones (thin black arrow). Conversely, the slides from DOX-treated rats revealed numerous highly branched brown stained astrocytes in the two inspected regions (thin black arrow). DOX + BISA50 showed fewer less branched astrocytes in both hippocampal regions (thin black arrow). DOX + BISA100 showed much fewer normal astrocytes in the two scrutinized hippocampal sections (thin black arrow). The brown areas (GFAP expression) were represented as optical density (OD). Data are reported as mean ± SEM (n = 6). Bars marked by different letters are significantly different (p < 0.05).
3.5 Impact of BISA on hippocampal apoptosis induced by DOXThe results in Figure 8 showed that the mRNA level the pro-apoptotic gene (Bax) increased significantly (p < 0.05) in the hippocampus of DOX-treated rats when compared to the controls. On the other hand, a substantial downregulation (p < 0.05) in the mRNA level of the anti-apoptotic gene (Bcl-2) was detected in these DOX-exposed animals as compared to the controls. Whereas, DOX + BISA groups exhibited a significant suppression (p < 0.05) in Bax mRNA and an elevation (p < 0.05) in the Bcl-2 transcription level in comparison with DOX-group. The transcription levels of these apoptotic markers were completely reversed in rats receiving DOX plus BISA at 100 mg/kg/day.
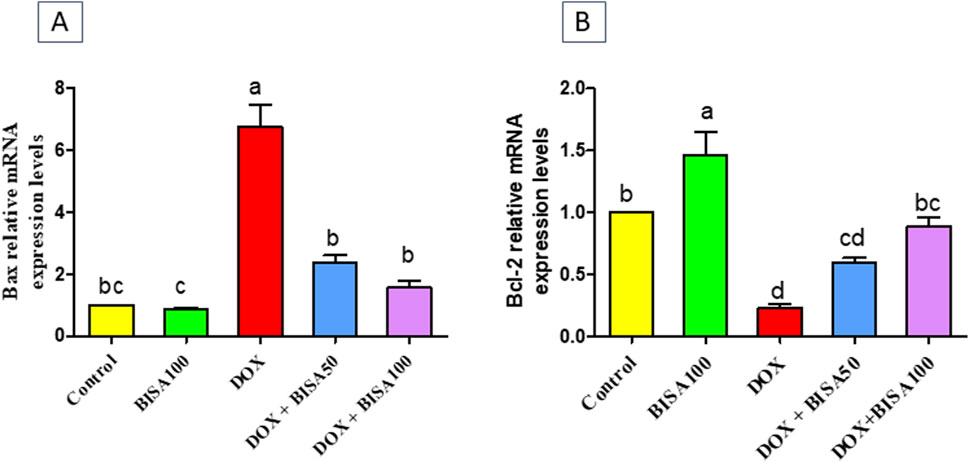
Figure 8. Effect of α-bisabolol on relative mRNA expression of apoptotic markers; (A) Bax and (B) Bcl-2 in the hippocampus of DOX-treated rats. Data are represented as mean ± SEM (n = 6). The lowercase letter on every bar clarify statistical difference among them (p < 0.05). BISA50, BISA100: α-Bisabolol was supplied by gavage at 50 and 100 mg/kg/day, DOX: doxorubicin was given i. p. Weekly for 4 weeks.
3.6 Effect of BISA on synaptic plasticity of the hippocampusTo assess the effect of BISA on the synaptic plasticity, we evaluated the concentrations of BDNF and its receptor TrKB in the hippocampal tissues utilizing ELISA. Th The DOX treatment caused a significant reduction (p < 0.05) in the levels of BDNF and TrKB by 74% and 57%, respectively, compared to the control group. Conversely, BISA co-treatment at 50 and 100 mg/kg daily exhibited a noticeable rise (p < 0.05) in the concentrations of BDNF and TrKB in relation to rats receiving DOX solely. The entire restoration to the control concentrations of BDNF and TrKB was accomplished only with BISA at 100 mg/kg (Figures 9A,B).
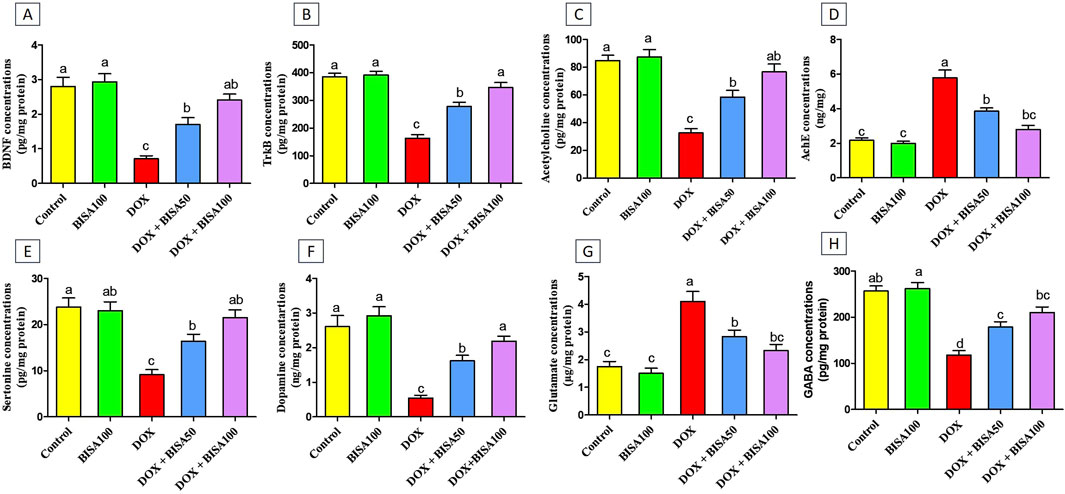
Figure 9. Effect of α-bisabolol on the protein expression of (A) BDNF, (B) TrKB, (C) Ach, (D) AchE, (E) 5-HT, (F) DA, (G) glutamate, (H) GABA in rat’s hippocampus in Dox-induced chemobrain. The findings are expressed as mean ± SEM (n = 6). Bars carrying various letters are substantially different (p < 0.05). BISA50, BISA100: α-bisabolol was introduced orally at 50 and 100 mg/kg/day, DOX: doxorubicin was injected i. p. Weekly for 4 weeks. BDNF: brain derived neurotropic factor, TrKB: tropomyosin receptor kinase B, ACh: acetylcholine, 5-HT: serotonin, DA: dopamine AChE: acetylcholinesterase, GABA: Gamma-aminobutyric acid.
Additionally, the concentrations of the neurotransmitters ACh, 5-HT, DA, and GABA were noteworthy decreased (p < 0.05) in the DOX group relative to the control one (Figures 9C,E,F,H). While, a substantial increase (p < 0.05) in AChE activity and glutamate level was recorded after DOX administration when compared to the control rats (Figures 9D,G). On the contrary, rats in the DOX + BISA50 and DOX + BISA100 groups displayed a marked elevation (p < 0.05) in ACh, 5-HT, DA, and GABA levels and a significant suppression (p < 0.05) in the activity of AChE and glutamate level in comparison with those in the DOX group. BISA at 100 mg/kg even returned their levels to near the control levels.
The qRT-PCR results demonstrated that the p38 MAPK mRNA level was remarkably higher (p < 0.05) in the hippocampus of rats treated with DOX only than the control rats. In contrast, both DOX + BISA groups markedly downregulated (p < 0.05) the mRNA expression of p38 MAPK compared to the DOX treatment alone. Furthermore, the transcription level of this gene returned to the normal in the DOX + BISA100 group (Figure 10).
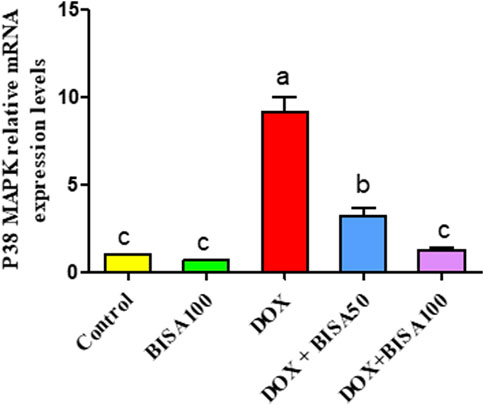
Figure 10. Influence of α-bisabolol on the relative mRNA expression of P38 MAPK in the hippocampus of rats exposed to DOX. The data are displayed as mean ± SEM (n = 6). Different letters on every bar point out statistical difference (p < 0.05). BISA50, BISA100: α-bisabolol was provided per os at 50 and 100 mg/kg/day, DOX: doxorubicin was given i. p. One injection per week for 28 days. P38 MAPK: P38 mitogen-activated protein kinase.
3.7 Impact of BISA on the mRNA levels of SIRT-1 in the hippocampusFigure 11 revealed a considerable downregulation (p < 0.05) in the transcription level of SIRT-1 in the DOX group in relation to control animals. Contrariwise, DOX + BISA50 and DOX + BISA100 groups showed a noticeable increase (p < 0.05) in the mRNA level of SIRT-1 gene counterweight to the DOX group. Notably, co-administration of BISA at 100 mg/kg with DOX revealed a better ameliorative action on SIRT-1 gene expression relative to the DOX + BISA50 group.
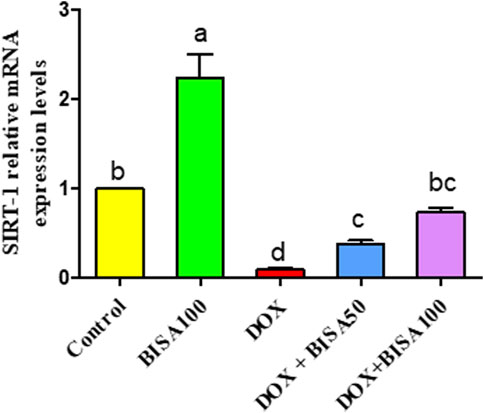
Figure 11. Impact of α-bisabolol on the relative mRNA expression of hippocampal SIRT-1 in DOX-induced cognitive impairment. Values are exhibited as mean ± SEM (n = 6). Bars with distinct lowercase letters are significantly different from one another (p < 0.05). BISA50, BISA100: α-bisabolol was gavaged at 50 and 100 mg/kg/day, DOX: doxorubicin was administered i. p. every week for 4 weeks. SIRT-1: sirtuin-1.
4 DiscussionThe present study aimed to assess the potential neuroprotective effect of BISA against DOX-provoked chemofog. The results of our study revealed that BISA exhibited a palliative role against cognitive disturbances caused by DOX in rats, as shown by the enhancement of short-term memory and spatial learning, restoration of the hippocamapal histological architecture, alleviation of the oxidative stress and neuroinflammation, suppression of apoptosis, and reversion of the synaptic dysplasticity. The higher dose of BISA (100 mg/kg/day) displayed a more remarkable neuroprotective effect against DOX-induced chemofog than the lower dose (50 mg/kg/day). Our findings are consistent with our hypothesis that BISA has a palliative effect on DOX-induced chemofog via its antioxidant, anti-inflammatory, and anti-apoptotic properties, and its effect on the BDN/TrKB pathway.
In the present work, i. p. injection of DOX at 2 mg/kg/week for 4 weeks resulted in memory and learning disorders, as demonstrated by considerable shortening in the time of step-down latency of rats in the passive avoidance test, as well as prolongation in the time of the escape latency and reduction in the duration spent by the rats in the platform quadrant in the MWM test. Prior investigations showed that treatment of rats with DOX led to amnesia and deteriorations in their spatial cognitive abilities (Christie et al., 2012; Mounier et al., 2021; Wahdan et al., 2020). Considering that the hippocampus is primarily responsible for memory accession, storage, and recalling (El-Agamy et al., 2018), in keeping with the neurobehavioral manifestation, microscopic examination of hippocampal tissues collected from rats receiving DOX only presented severe neuronal degeneration. Meanwhile, the concurrent treatment with BISA alleviated the cognitive decline caused by DOX in both the behavioral and histoarchitectural conditions. In agreement with our results, Nazarinia et al. (2023) reported that oral BISA at 100 mg/kg/day for 10 days caused neurological protection and mitigated the memory impairment in rats experiencing seizures induced by pentylenetetrazole (PTZ).
The oxidative stress may be a major culprit in cognitive disorders caused by DOX. the quinone-containing compound (Joshi et al., 2010). Du et al. (2021) reported that the oxidative insult causes DOX-provoked cognitive dysfunction via direct and indirect mechanisms. DOX goes through a redox cycling process that involves the incorporation of a single electron to the quinone portion, producing a semi-quinone structure that is subsequently transformed back to the original quinone with the consequence of generating large quantity of reactive oxygen species (ROS) which harms both the quality of structure and performance of multiple crucial biological molecules, including lipids, proteins, and nucleic acids (Keeney et al., 2018). It has been reported that DOX-triggered oxidative insult causes elevation in lipid peroxidation and protein oxidation along with reduction in the concentrations of enzymatic and nonenzymatic antioxidants (Keeny et al., 2018). Previous studies revealed that DOX can evoke direct neuronal damage by enhancing the generation of ROS and the depolarization of mitochondrial neurons in hippocampal neurons (Du et al., 2021). Despite ROS is believed to perform an important role in triggering the neurotoxicity of DOX, the above mentioned theory is not reliable for introducing ROS into the CNS since DOX cannot penetrate the BBB (Bigotte et al., 1982). Hence, this suggests the probable involvement of an indirect process that does not include redox reactions in the brain. In parallel, Trachootham et al. (2008) demonstrated that free radicals produced from redox cycling enhance NF-κB, a redox-responsive transcriptional factor, with a subsequent stimulation of the expression of several inflammatory mediators, including TNF-α, IL-6, and IL-1β. Among these mediators of inflammation, TNF-α can pass the BBB and activate astrocytes, the main component of microglia, and are implicated in the synaptic transmission (Kimelberg and Nedergaard, 2010). Baune et al. (2012) revealed that astrocytes play a crucial role in the neuroinflammatory process as they react to inflammatory mediators’ insults inducing astrogliosis, a condition where the action of astrocytes is reversed to be deleterious and is associated with an excessive liberation of inflammatory signals. This results in a cycle of detrimental neurological inflammation that boosts the nitrosative and oxidative stresses and lowers synaptic plasticity. In this regard, inflammation has been considered to be another essential triggering factor, together with the oxidative insult, with cytokines representing an additional incorporating culprit in the progression of cognitive dysfunction caused by DOX (Aluise et al., 2010; El-Agamy et al., 2018).
In our research, the memory impairment caused
Comments (0)